Drs. Colleen A. McClung and Yanhua Huang Find That Nucleus Accumbens Excitatory Signaling Is Integral to Effects of Clock Gene Disruption on Manic-like Behaviors
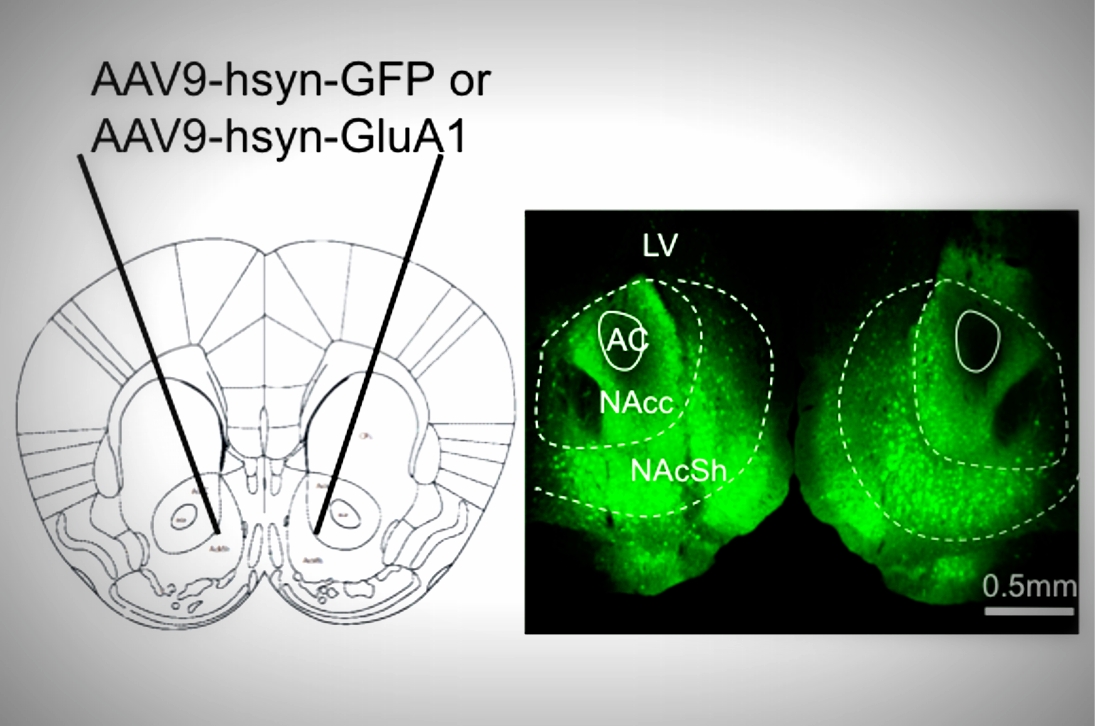
Disruptions in circadian rhythms are associated with an increased risk for bipolar disorder, and the circadian protein CLOCK is involved in regulating monoaminergic systems and mood-related behavior. Colleen A. McClung, PhD, Professor of Psychiatry and Yanhua Huang, PhD, Assistant Professor of Psychiatry set out to clarify the molecular and synaptic mechanisms underlying this relationship. Working in collaboration with McClung Lab student researchers Puja K. Parekh, Darius Becker-Krail, and Poornima Sundaravelu, they characterized alterations in excitatory synaptic transmission, strength, and intrinsic excitability of nucleus accumbens (NAc) neurons. Using ex vivo whole-cell patch-clamp electrophysiology in ClockΔ19 mutant and wild-type mice, they performed protein crosslinking and western blot analysis to examine surface and intracellular levels and rhythm of the glutamate receptor subunit, GluA1, in the NAc. They also used viral-mediated overexpression of Gria1 in the NAc and behavioral assays.
Compared with wild-type mice, ClockΔ19 mice displayed reduced excitatory synaptic responses at NAc medium spiny neurons (MSNs). These alterations are likely postsynaptic, as presynaptic release of glutamate onto MSNs is unaltered in mutant mice. Additionally, NAc surface protein levels and the rhythm of GRIA1 are decreased in ClockΔ19 mice diurnally, consistent with reduced functional synaptic response. The investigators also observed a significantly hyperpolarized resting membrane potential of ClockΔ19 MSNs, suggesting lowered intrinsic excitability. Importantly, overexpression of functional Gria1 in the NAc of mutant mice normalized increased exploratory drive and reward sensitivity behavior when mice were in a manic-like state.
Collectively, the study findings support a hypothesis that diminished CLOCK protein function increases dopaminergic activity and tone, altering excitatory drive onto MSNs. This may reduce the functional output of the NAc in feedback dynamics onto the ventral tegmental area or disinhibit other target regions, leading to the elevation of mood, exploratory drive, and reward-seeking behavior associated with bipolar mania. “These results help us to better understand the strong relationship between circadian rhythm disruption and bipolar disorder and the mechanisms by which circadian proteins regulate activity in regions of the brain associated with mood and reward,” said Dr. McClung.
Altered GluA1 (Gria1) Function and Accumbal Synaptic Plasticity in the ClockΔ19 Model of Bipolar Mania
Parekh PK, Becker-Krail D, Sundaravelu P, Ishigaki S, Okado H, Sobue G, Huang Y, McClung CA
Published online in Biological Psychiatry, 2017,
